|
For acidic amino acids, the isoelectric point will be at lower pH as the acidic side chain will introduce an extra negative charge and for basic amino acids, the isoelectric point will be at higher pH as the basic side chain will introduce an extra positive charge. For neutral amino acids, the side chains are neutral and the isoelectric point is given simply by the average of the pK a values of carboxylic acid and amine. We used the SARSCoV2 proteins currently available on the market, namely the RBD with His and Fctag, the S1 subunit with Histag, and the S1/S2 subunits with Histag. and Colin Grantham, Online Stator and Rotor Resistance Estimation. This information has been sourced, reviewed and adapted from materials provided by. The Multi Purpose Titrator MPT2 used in conjunction with a Zetasizer Nano instrument allows for the automatic determination of isoelectric points. Note: The isoelectric point is given by the average of the pK a values that involve the zwitterions, not just by the pK a values that describe the carboxylic acid group and the amine group. Since the isoelectric point of proteins is an important property, we sought to determine it using the Maurice (imaged) CIEF system from ProteinSimple, a BioTechne brand. (PI), and Proportional Integral Derivative(PID) controller for speed control of. The determination of isoelectric points is important in understanding the stability of colloidal dispersions. The ability to conduct measurements in free solution thus provides the basis for the rapid determination of isoelectric points of proteins under a wide variety of solution conditions and in small volumes.Since the isoelectric point is given by the average of the pK a values that involve the zwitterion, so we can write the formula for lysine as: We will also discuss zwitterions, or the forms of amino acids that dominate at the isoelectric point. By definition, the aqueous solution of an ampholyte is in the isoelectric point if exactly the same amount of hydrogen ion is dissociated (usually from carboxyl. The pH values of solutions of gelatin in water approached the limit 4.86 +/- 0. The isoelectric point of this material was determined in four ways. As a result, application of this precise and rapid determination method enables us to reasonably regulate the pI and surface charge of chrome-free leather so as. You will learn how to calculate the isoelectric point, and the effects of pH on the amino acid's overall charge. Two samples of a standard gelatin were studied, both prepared according to published specifications and washed free from diffusible electrolytes. To demonstrate the general approachability of this platform, we have measured the isoelectric points of representative set of seven proteins, bovine serum albumin, β-lactoglobulin, ribonuclease A, ovalbumin, human transferrin, ubiquitin and myoglobin in microlitre sample volumes. The isoelectric point of an amino acid is the pH at which the amino acid has a neutral charge. Online calculation (prediction) of theoretical isoelectric point (pI, IEP) of proteins and petides from sequence alone. 22. In particular, in this approach, the pH of the electrolyte solution is modulated in time rather than in space, as in the case for conventional determinations of the isoelectric point. 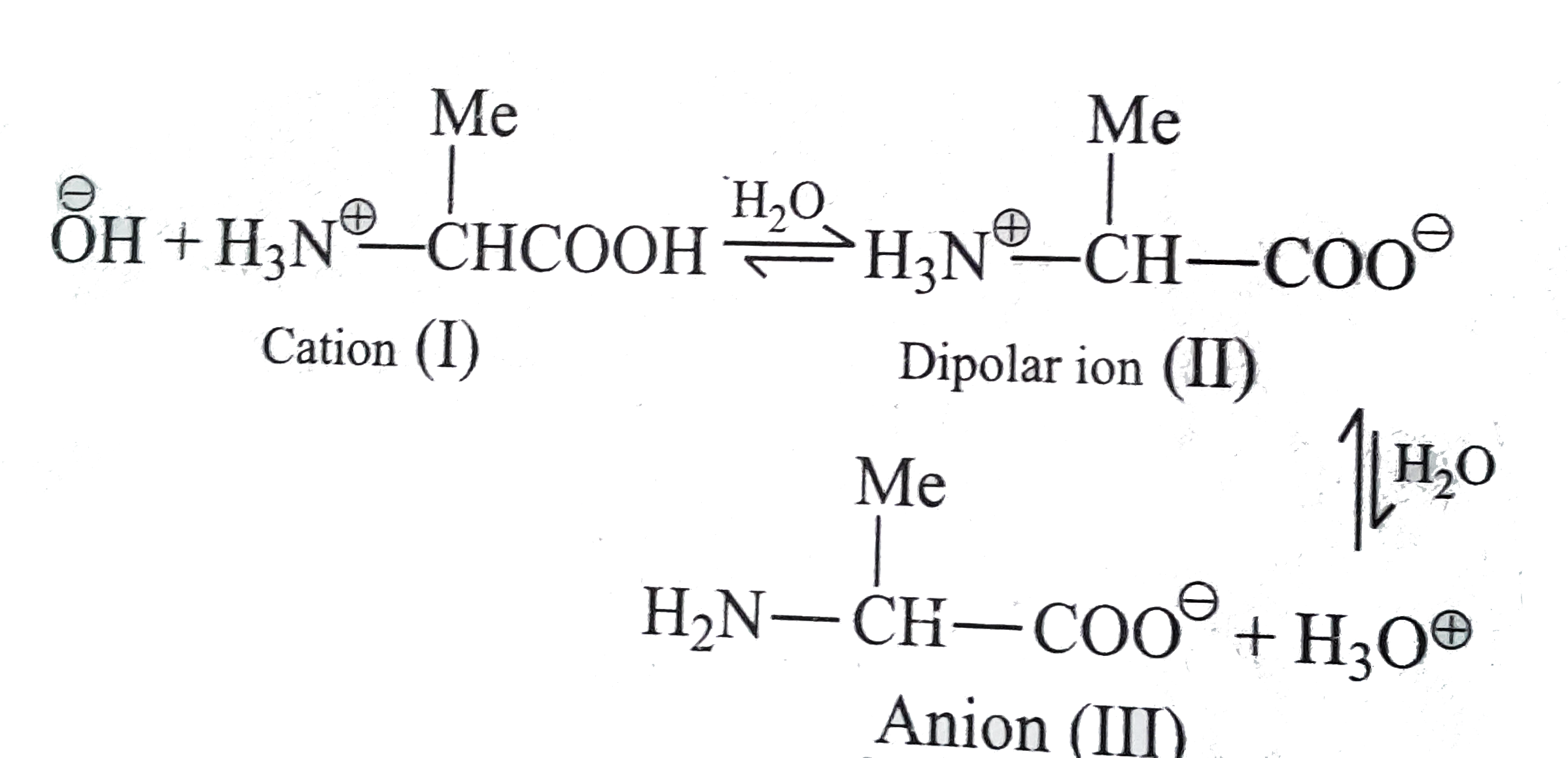
Here, we introduce a gradient-free approach, exploiting a microfluidic platform which allows us to perform rapid pH change on chip and probe the electrophoretic mobility of species in a controlled field. The majority of conventional methods for the determination of the isoelectric point of a molecule rely on the use of spatial gradients in pH, although significant practical challenges are associated with such techniques, notably the difficulty in generating a stable and well controlled pH gradient. The majority of conventional methods for the determination of the isoelectric. 
The isoelectric point (pI) of a protein is a key characteristic that influences its overall electrostatic behaviour. The isoelectric point (pI) of a protein is a key characteristic that influences its overall electrostatic behaviour. The isoelectric point of mAb A is above pH 9, and the isoelectric point of mAb B is below pH 8 (relative difference between them is 1.5 units), which again puts both mAbs on the opposite sides of. The isoelectric point (pI) of a protein is a key characteristic that influences its overall electrostatic behaviour.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
